Loading... Please wait...
Loading... Please wait...Categories
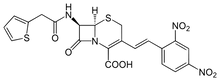
Nitrocefin
Purity >95.0%
| CAS Number: | 41906-86-9 |
| Chemical Formula: | C21H16N4O8S2 |
| Molecular Weight: | 516.5 g/mol |
Nitrocefin is a cephalosporin with chromogenic properties and is routinely used to detect beta-lactamase enzymes produced by beta-lactam resistant bacteria. Nitrocefin is soluble in DMSO and is commonly used at a 1.0 mg/mL concentration.
Essentially all beta-lactamase enzymes hydrolyze the amide bond between the carbonyl carbon and the nitrogen in the beta-lactam ring of nitrocefin. Macroscopic detection of this process is made possible because a ultraviolet absorption shift from intact versus hydrolyzed nitrocefin occurs within the visible light spectrum (~380 nm to ~500 nm, or yellow to red).
Nitrocefin Protocols:
The chromogenic properties of Nitrocefin degradation can be exploited in several simple and rapid assays for detection of β-lactamases. This protocol will outline preparation of Nitrocefin solution and its use in β-lactamase detection methods.
Working solution concentrations of Nitrocefin typically are within a range of 0.5 mg/ml–1.0 mg/ml and appear as a yellow color after preparation.
We recommend preparing a concentrated stock solution of 10 mg/mL in DMSO. The proceeding example will describe how to prepare a stock solution (10.0 mg/mL) and a working concentration (1.0 mg/mL) using 10.0 mg of Nitrocefin.
Stock solution–10.0 mg/mL in DMSO
a. Add 10.0 mg Nitrocefin powder to 1.0 mL DMSO in appropriate container. (If the container only has 10.0 mg Nitrocefin, it may be more efficient to add the DMSO directly to the container, vortex the contents, and add contents to an appropriate container.)
Note: Store any remaining stock solution or working solution at -20°C and protect from light.
Working solution–1.0 mg/mL in PBS
a. Add 9.0 mL PBS buffer (100mM, neutral pH) to the prepared stock solution to bring the final volume to 10.0 mL
Note: Working solution should be used within 14 days after preparation.
If the solution at this concentration produces a red color before exposure to β-lactamase, dilute the working solution 10X or more until a yellow color is achieved.
Rapid Detection Assays:
The following assays should produce a positive result within moments after contact with β-lactamase producing organisms.
However, it is recommended to allow 20-30 min. before concluding with a negative result.
Slide Surface Assay
1. Add one drop of 1.0 mg/ml Nitrocefin to the surface of a clean glass slide.
2. Select a colony from an agar surface us ing a sterile loop and mix with the drop.
3. Appearance of red color within 20-30 min. indicates β-lactamase activity.
Note: If needed, add additional Nitrocefin solution to prevent drying.
Direct Contact Assay
1. Place one drop of 1.0 mg/ml Nitrocefin directly on the surface of an isolated colony.
2. Appearance of red color within 20-30 min. indicates β-lactamase activity.
Broth Suspension Assay
1. Add 3-5 drops of 1.0 mg/ml Nitrocefin to 1 ml of broth suspension.
2. Appearance of red color within 20-30 min. indicates β-lactamase activity.
Lysed Cell Assay
1. Lyse 1ml of cell suspension by sonication.
2. Add 3-5 drops of 1.0 mg/ml Nitrocefin to lysed cell suspension.
3. Appearance of red color within 20-30 min. indicates β-lactamase activity.
Filter Paper Assay
1. Place a small piece of filter paper (~3 x 3 cm) in a clean petri dish or another clean isolated surface and saturate (3-5 ml) with 1.0 mg/ml Nitrocefin
2. Select an isolated colony and smear over the surface of the impregnated filter paper.
3. Appearance of red color within 20-30 min. indicates β-lactamase activity.
Solution preparation and color change before and after β-lactamase exposure

(A) Concentrated nitrocefin (10.0 mg/mL) in DMSO before dilution with PBS buffer.
(B) Nitrocefin diluted with PBS buffer to working concentration (1.0 mg/mL). The yellow color is indicative of intact, undegraded nitrocefin.
(C) 25 units of beta-lactamase dropped on top of nitrocefin (1.0 mg/mL in PBS). The red color is the result of beta-lactamase mediated cleavage of the nitrocefin.
(D) Vortexed mixture of contents shown in picture (C).
Spectrophotometric Analysis of Nitrocefin Degradation
In aqueous solution, at approximately neutral pH, nitrocefin produces two absorption peaks: one at 217 nm which corresponds to the 7-acyl group and another at 386 nm. After exposure to beta-lactamase, the peak at 386 nm diminishes, and a new peak appears at 482 nm which corresponds to degraded nitrocefin.
This shift in absorption can be measured over time which enables a scientist to measure the activity of a given beta-lactamase enzyme based on the rate at which it degrades nitrocefin.
The following equations were proposed by O’Callaghan et al. (see references).
1. Enzyme activity (pH 7.0, 37°C, 386nm) = (x/1.03) · y · U
2. Enzyme activity (pH 7.0, 37°C, 482nm) = (x/1.59) · y · U
Activity is defined as μmol substrate destroyed per minute per ml of enzyme
X = ΔOD/minute
Y = number of μmol nitrocefin
U = rate at which nitrocefin is degraded by a given enzyme (Michaelis constants (Km) and substrate specificity are determined by Line-weaver-Burk plots).
1.03 in eqn.1 is equal to the ΔOD (1.75 to 0.72) at 386nm
1.59 in eqn. 2 is equal to the ΔOD (0.15 to 1.74) at 482nm
Celebrity Endorsements
Parr, T. R., Jr. "Simple Screening Method for Beta-lactamase-positive and -negative Ampicillin-resistant Haemophilus Influenzae Isolates." Journal of Clinic al Microbiology 20.1 (1984): 131-32.