Loading... Please wait...
Loading... Please wait...Categories
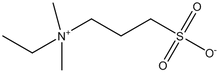
NDSB 195
(NDSB-195; Dimethylethylammonium propane sulfonate; Non-detergent sulfobetaines 195)
Purity > 99.0 %
| CAS Number: |
160255-06-1
|
| Chemical Formula: |
C8H18N2O4S
|
| Molecular Weight: |
195.3 g/mol
|
NDSB 195 is a zwitterionic compound that is used in protein crystallization and refolding. NDSB 195 is able to prevent protein aggregation and facilitate the renaturation either chemically and thermally denatured proteins. The applications are wide ranging in protein biochemistry useful to enhance extraction, solubilization and crystallization. NDSB is very soluble in water (greater than 2.0 molar) and do not alter significantly the pH or viscosity of biological buffers.
NDSB 195 does not show absorbance at 280 nm which is convenient for measuring protein concentration. In addition, enzymes have been shown to remain active in the presence of this reagent and can easily be removed by dialysis.
Free Shipping within the Continental USA
Celebrity Endorsements
-This paper shows a significant increase in the yield of protein extraction by the addition of sulfobetaine-type mild solubilization agents in membrane proteins and proteins from lyophilized platelets, with a maximum increase in yield of up to 100%.
Klammt, C.; et. al. (2004) High level cell free expression and specific labeling of integral membrane proteins. European Journal of Biochemistry. Vol 271, 568–580.
-Examines a large range of detergents including NDSBs. Overall, the paper demonstrates the high level expression of integral membrane proteins in a cell-free coupled transcription/translation system using a modified Escherichia coli S30 extract preparation and an optimized protocol.
Vincentelli, R.; et. al. (2004) High throughput automated refolding screening of inclusion bodies. Protein Science. 13(10). 2782–2792.
-Examines the expression of foreign proteins in Escherichia coli is the occurrence of amorphous aggregates of misfolded proteins, called inclusion bodies. Developing efficient protein native structure recovery procedures based on protein refolding is therefore an important challenge. This paper examines various buffers that can be used for the refolding of inclusion bodies.
Vuillard, L.; et. al (1996) Protein crystallography with non-detergent sulfobetaines. Journal of Crystal Growth. Vol 168, 150-154.
-This manuscript examines protein crystal growth of described non-detergent solubilizers/stabilizers sulfobetaines (NDSB). Lysozyme was used as the model protein which was examined by X-ray crystallographic methods. NDSB resulted in the increase in protein solubility and the crystal growth rate without influencing the quality of crystals as determined by comparing Wilson plots. Sulphur anomalous X-ray scattering was used to show the absence of any bound NDSB in the crystals. NDSB was shown to be critical in solving the structure of Desulfovibrio Gigas type II ferrodoxin since it showed a reduction in crystal twinning and resulted in the growth of a new crystal form. This papers sheds light on the unique combination of solubilizing and stabilizing properties of NDSB molecules as valuable additives for protein crystal growth.