Loading... Please wait...
Loading... Please wait...Categories
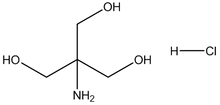
Tris HCl
(Tris(hydroxymethyl)aminomethane Hydrochloride; 2-Amino-2-(hydroxymethyl)-1,3-propanediol hydrochloride; Tris (hydroxymethyl) aminomethane Hydrochloride)
Purity > 99.0%
| CAS Number: |
1185-53-1
|
| Chemical Formula: |
C4H11NO3·HCl
|
| Molecular Weight: | 157.59 g/mol |
Tris hydrochloride (Tris Cl) is a buffer used throughout scientific research. Tris Cl is a Good Buffer with an effective pH range between 7.0 and 9.2. The pKa of Tris 8.0 therefore it has limited buffer capacity outside the range of 7.5 to 9.0. This pH range is often targeted in biological systems which lead to its common usage.
Free Shipping within the Continental USA
Protocol for the Preparation of 1.0 M Tris HCl Stock Solution:
1) Dissolve 121.1 g of Tris base in 600 ml of H2O
2) Add the appropriate about of HCl to achieve your desired pH
| Desired pH (25° C) |
Volume 0.1 N HCl (ml)
|
| 7.20 |
44.7
|
| 7.50 | 40.3 |
| 8.00 | 29.2 |
| 8.50 | 14.7 |
3) Adjust the volume to 1 L with H2O
The pH of Tris HCl is quite temperature dependent. The pH of a Tris buffered solution decreases about 0.03 units for each 1 degree Celsius increase in temperature.
Tris HCl can also be found in HPLC protocols as well as a buffer in protein crystallization solutions.
Comparable Items:
| Alfa Aesar | A11379-18 |
| Alfa Aesar | A11379-30 |
| Alfa Aesar | A11379-0B |
| Amresco | 0234-500G |
| Amresco | M108-1KG |
| Amresco | 0234-5KG |
| Amresco | E553-500ML |
| Amresco | 0234-12KG |
| Amresco | 0234-1KG |
| Amresco | E553-100ML |
| Avantor | 4103-02 |
| Avantor | 4103-04 |
| Avantor | 4103-06 |
| Avantor | 4103-07 |
| Avantor | H590-88 |
| Avantor | 4106-09 |
| Avantor | 4106-07 |
| Avantor | 4106-01 |
| Avantor | 4103-01 |
| Avantor | 4106-05 |
| EMD Millipore | 9310-500GM |
| EMD Millipore | 9350-5KG |
| EMD Millipore | 9380-25KG |
| G-Biosciences | RC-108 |
| G-Biosciences | RC-107 |
| HiMedia | M108-500G |
| JT Baker | 4103-F7 |
| JT Baker | 4106-F9 |
| MP Biomedicals | 816100 |
| MP Biomedicals | 816116 |
| MP Biomedicals | 816124 |
| TCI America | T0740-25G |
| TCI America | T0740-500G |
| United Biochemicals | 85855 |